Repeated Measures
Easy-to-use platform combining low patient burden with high reliability and patient adherence.
Neulogiq provides frequent, reliable in-clinic and at-home longitudinal monitoring of discrete brain functions.
Discover
A unified platform integrating tablet-based assessments, longitudinal EEG, and real-world data to capture objective neurological change.


Easy-to-use tools that fit naturally into daily life.
Reliable, repeatable brain function data you can trust.

In-clinic and at-home assessments, unified in one platform.

Easy-to-use measurement tools that patients want to engage with.

Reliable, repeatable brain function data you can trust.

Engaging multi-modal assessments for use in-clinic or at-home, unified in one platform.
Reliable, longitudinal insights collected in-clinic and at home.
Easy-to-use platform combining low patient burden with high reliability and patient adherence.
Incorporate data from multiple modalities such as cognition, EEG, mood, language and sleep, simultaneously.
Built on our reproducible, reliable analysis pipeline using advanced signal processing and machine learning to characterize EEG signals.
The NeuLogiq platform measures multiple domains of brain function to provide a comprehensive view of CNS health and treatment response.
Tablet-based cognitive assessments designed for repeated, real-world use.
Neurophysiological measurements capturing brain network activity, synchronised with behavioural data.
Assessment of emotional processing and mood-related signals to detect changes over time.
Analysis of natural speech, vocabulary, organization and prosody to monitor language and cognitive function.
Remote measurement of sleep quality and sleep staging using validated digital tools.
UKCA-marked and FDA 510(k) cleared medical device, combining regulatory compliance with award-winning design for clinical and research EEG applications.

Comprehensive, multi-modal neuro-assessments for both in-clinic and at-home measurement of cognition, neurophysiology and behaviour. Grounded in scientific literature, repeatable and engaging, language-agnostic and suitable for deployment at scale globally.

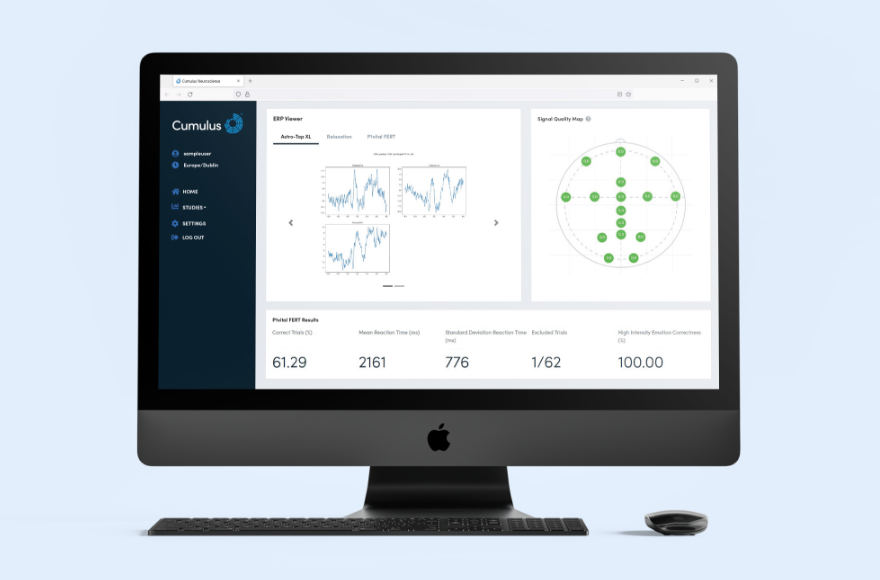
Proprietary machine learning-based methods are used to evaluate the multi-dimensional real-world datasets generated through NeuLogiq. These analytics are designed to support exploratory research analysis, enabling the identification of subtle patterns and changes within datasets to inform research hypotheses and trial insights.

Automated, AI-powered dashboards for viewing incoming data from distributed locations across the globe. Provides rapid access to assist in monitoring compliance and data quality of decentralized measurements.

UKCA-marked and FDA 510(k) cleared medical device, combining regulatory compliance with award-winning design for clinical and research EEG applications.

Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Ut enim ad minim veniam, quis nostrud exercitation ullamco laboris nisi ut aliquip ex ea commodo consequat.

Proprietary machine learning-based methods are used to evaluate the multi-dimensional real-world datasets generated through NeuLogiq. These analytics are designed to support exploratory research analysis, enabling the identification of subtle patterns and changes within datasets to inform research hypotheses and trial insights.

Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Ut enim ad minim veniam, quis nostrud exercitation ullamco laboris nisi ut aliquip ex ea commodo consequat.











Here you can find answers to common questions.
Review our publications, posters, video interviews and webinars featuring biopharma customers
The NeuLogiq® Platform can be used across the full clinical development lifecycle from small Phase 0 to large Phase 3 studies.
Our team has experience deploying the NeuLogiq platform in the in the UK, US, Europe, Australia, Canada and Japan, with language support and scalability built-in.
Our Clinical Operations and Scientific teams work with biopharma sponsors and clinical sites to provide end-to-end operational, technical and scientific support from trial design through deployment and analysis.
Yes. We can support studies in AD that include participants with mild cognitive impairment. Our CNS-101 observational study included 59 people with dementia and 60 aged-matched controls, and showed 80% adherance to the study protocol. See the CNS-101 data presented at CTAD 2025.
No. Cumulus Neuroscience deploys the NeuLogiq Platform solely for use in Clinical Studies. We do not sell the NeuLogiq Platform for individual use.